The functional group approach " works" because the properties and reaction chemistry of a particular functional group (FG) can be remarkably independent of environment. An inert hydrocarbon skeleton onto which functional groups (FGs) are attached or superimposed.Organic chemistry is dominated by the "functional group approach", where organic molecules are deemed to be constructed from: Briefly, the sequence rules rank the substituents in order of decreasing atomic number and if two or more atoms connected to the C atom are the same the second atom determines the order. The other isomer is E (for e ntgegen meaning opposite). If the two with the higher rankings are on the same side of the double bond, that isomer is Z (for the German word zusammen, which means together). The newer method ranks the substituents for each C atom according to the Cahn-Ingold-Prelog sequence rules. But it won’t work well if there were four different atoms involved.
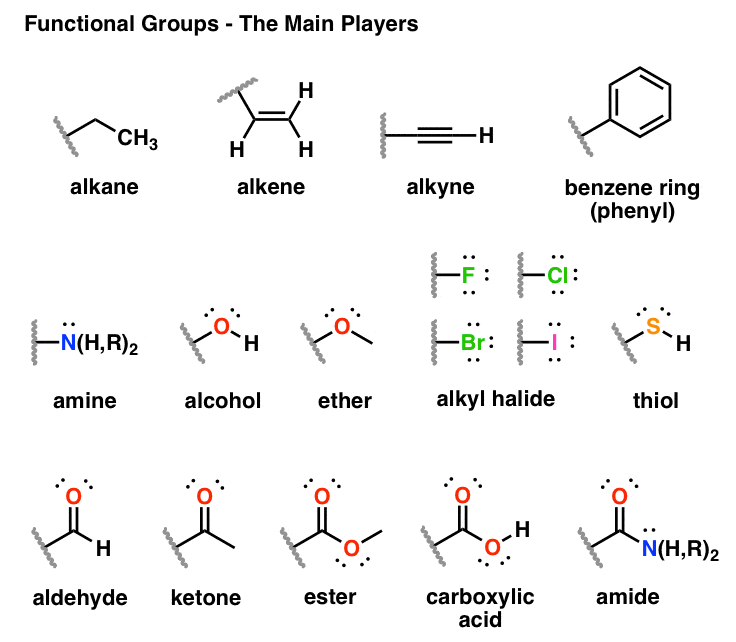
The older method uses cis- and trans- which works well for the example given above because there are two H atoms and two Cl atoms. The isomers are diasteromers according to the definition given here. These isomers are superimposable on their mirror images if no chiral centers are present. Geometric isomers are generally not optical isomers unless they also happen to have chiral centers. Geometric isomers are stereoisomers that are distinct and separate because they cannot freely rotate due to a multiple bond or a ring structure. The table below summarises all the tests and the chemical reactions that take place.Stereoisomers (briefly and non-optical only!) Tollens’ reagent or Fehling’s solution can then be used to determine if the initial alcohol was primary or secondary. A primary alcohol will form an aldehyde and a secondary alcohol will form a ketone. If no colour change is observed then the alcohol was a tertiary alcohol. If the potassium dichromate solution changes colour from orange to green then an oxidation reaction has taken place with a primary or secondary alcohol. The mixture is heated gently followed by immediate distillation of the product. The alcohol is mixed with a small quantity of potassium dichromate and sulphuric acid in a pear shaped flask. The oxidising agent potassium dichromate dissolved in dilute sulphuric acid will oxidise primary and secondary alcohols but not tertiary alcohols. Test for alcoholsĪlcohols are classified as being primary, secondary or tertiary depending upon how many alkyl or aryl groups (commonly known as “R” groups) are attached to the carbon atom that is attached to the hydroxy group.Ī Level Chemistry students are required to distinguish between primary, secondary and tertiary alcohols. Tollens’ reagent (known as ammoniacal silver nitrate) when heated with aldehydes in a boiling tube forms a silver mirror on the inside of the tube. There are two chemical tests that will give a positive test for an aldehyde but not a ketone.įehling’s solution (a mixture of solutions of copper sulphate and sodium hydroxide) is a blue solution which when gently heated with an aldehyde forms a red precipitate of copper(I) oxide. The A Level syllabus requires candidates to be able to carry out a chemical test to distinguish an aldehyde from a ketone. It is very important that any unreacted hydroxide ions are removed completely to give a neutral or acidic solution as silver hydroxide will also produce a precipitate. Chloride ions will produce a white precipitate, bromide a cream precipitate and iodide produces a yellow precipitate. The halide ions can be detected using nitric acid followed by silver nitrate. Gentle heating will produce an alcohol and liberate halide ions. A small quantity of ethanol is also required to dissolve the haloalkane. The presence of a halogenoalkane (except fluoroalkanes) can be confirmed by first carrying out a nucleophilic substitution reaction with aqueous sodium hydroxide solution. Effervescence will indicate the production of a gas and bubbling it through limewater will confirm that the gas is carbon dioxide. Sodium carbonate is as good a choice as any. Test for carboxylic acidsĬarboxylic acids will react with metal carbonates to produce a salt, water and carbon dioxide. Bromine water is orange but the halogenoalkane formed will be colourless.

Test for alkenesĪlkenes will react with bromine water to form a halogenoalkane. Mechanisms for some of the reactions listed below can be found here. If you have not already done so, you should read my previous post and ensure that you are familiar with the chemical tests listed here.

In this blog post I summarise all the organic tests required for A Level Chemistry.
